People use the phrase "dopamine addiction" because it feels like a clean explanation for compulsive behavior. Someone cannot stop scrolling, gaming, gambling, using porn, shopping, or taking a substance, so the conclusion seems obvious: they must be addicted to dopamine.
That phrase points toward a real problem, but it is not the most accurate way to describe what is happening.
You are not literally addicted to dopamine the way a person can become dependent on a drug. Dopamine is a normal neurotransmitter your brain uses every day. What people usually mean by "dopamine addiction" is that they feel trapped in a reward-seeking loop: certain cues grab their attention, certain behaviors feel urgent, and the pattern keeps repeating even when the payoff is getting weaker [1]-[5].
That is a more useful place to start.
The phrase survives because it matches a real experience. People notice that:
So they reach for the simplest story available: dopamine must be the addictive thing.
The trouble is that this collapses several different processes into one word. It confuses a normal brain messenger with the larger reward-learning system that decides what to notice, what to chase, and what to repeat [3]-[5], [10], [13].
Dopamine is part of the brain's reward system, but it is not just a "pleasure chemical." That shortcut is too simple.
According to the National Institute on Drug Abuse, dopamine helps reinforce behaviors that the brain marks as important. A burst of dopamine signals that something meaningful just happened and that it may be worth repeating. Over time, that helps build habits [1], [2].
That matters because the brain does not only learn from the reward itself. It also learns from the cues around the reward:
This is one reason urges can feel powerful before the reward even arrives. The brain has already started predicting what comes next.
In other words, dopamine is less useful as a symbol for raw pleasure and more useful as part of a system for motivation, reinforcement, and learning [3], [4].
The tighter scientific version of that idea is reward prediction error. Keiflin and Janak show how midbrain dopamine neurons respond to surprise: unexpected reward increases firing, expected reward produces less response, and omitted reward pushes activity below baseline. That makes dopamine a teaching signal, not a pleasure meter [5]. Baik's receptor-level review adds the next layer: D1-like and D2-like receptors do not simply mirror each other, so "dopamine" is never one uniform process [4]. The brain is learning what to predict, what to pursue, and what to repeat, not just registering how good something feels [5], [10], [13].
Later syntheses caution that the dopamine story is broader than any single receptor family, brain region, or theory snapshot [12], [15], [16].
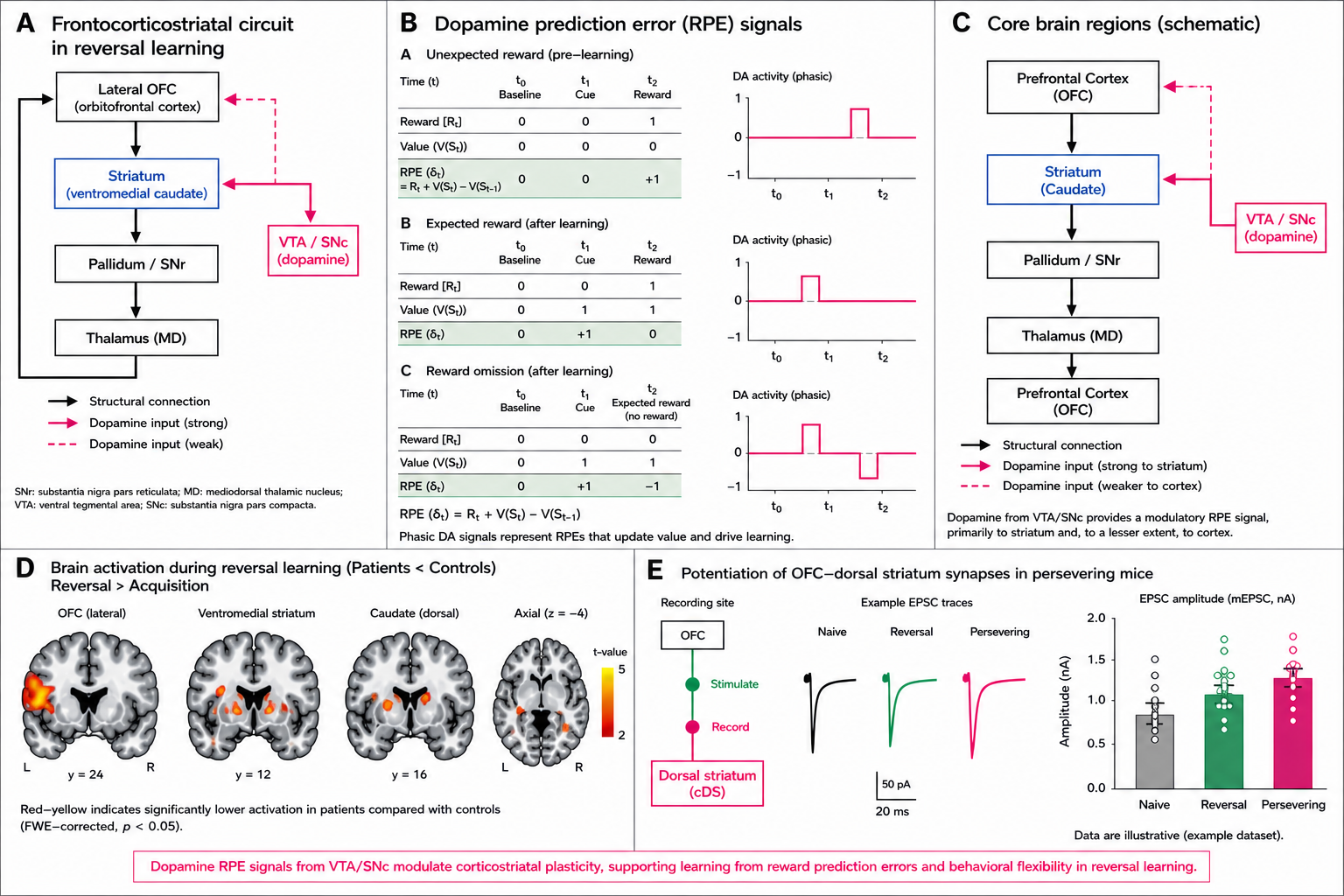
This first figure is a circuit diagram and prediction-error summary. Panel A shows the frontocorticostriatal loop, with the orbitofrontal cortex, striatum, pallidum, thalamus, and prefrontal cortex connected by structural arrows and dopamine input from VTA/SNc. Panel B shows that unexpected reward produces a dopamine spike, expected reward moves the signal to the cue, and reward omission produces a dip.
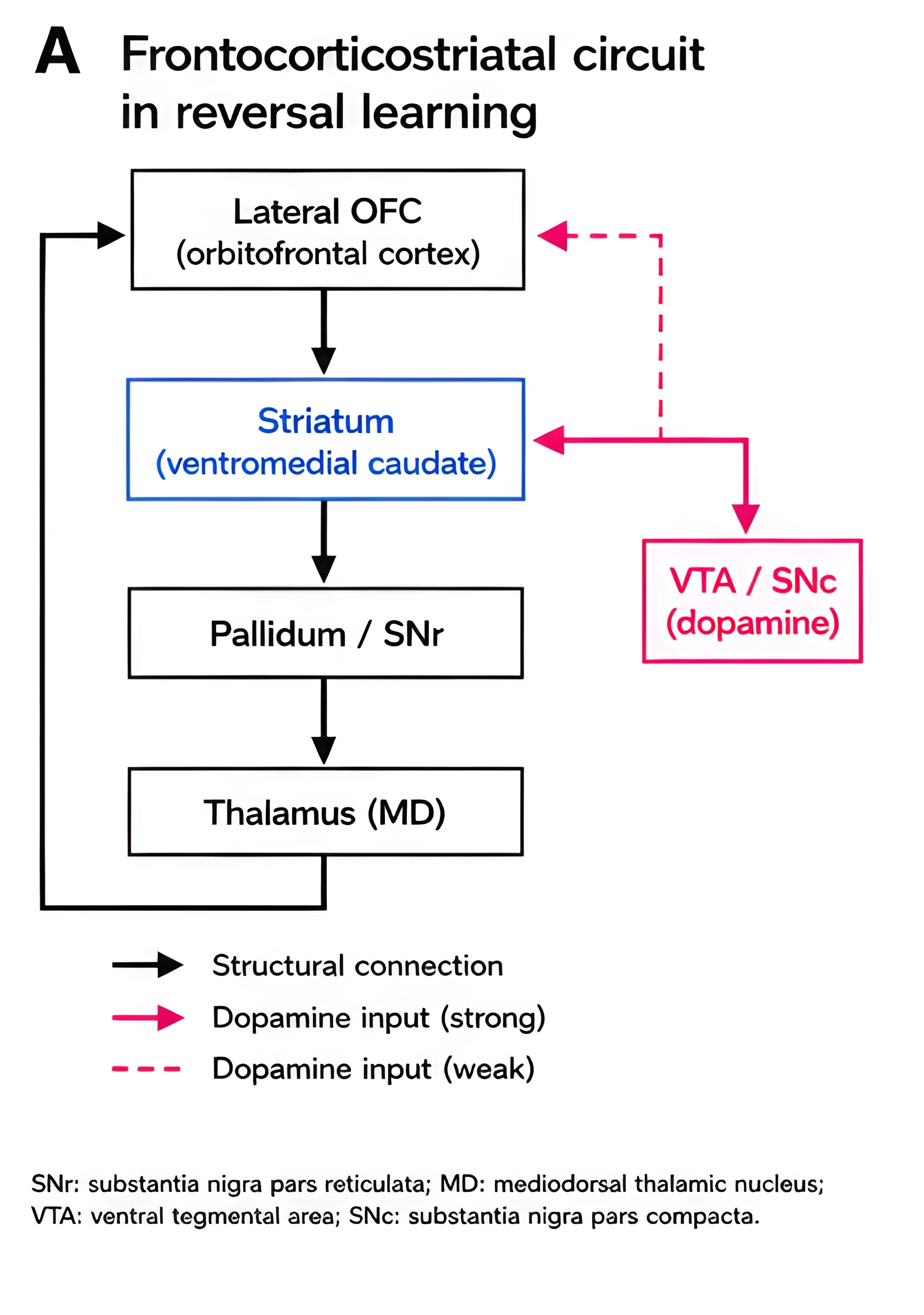
That is why the same neurotransmitter can support both ordinary learning and pathological repetition. A person is not "addicted to dopamine" in the literal sense. They are caught in a learned reward model that keeps updating in the direction of the cue, the ritual, and the expected payoff [5], [10], [13].
Once the brain learns a reward pattern, the cue can become as important as the reward itself. That is the core of cue-reactivity and it shows up well beyond substances. Starcke and colleagues' meta-analysis of behavioral addictions found stronger cue responses in gambling, gaming, and buying disorders across subjective, physiological, and neural measures [6]. Another meta-analysis on drug, gambling, food, and sexual cues found substantial overlap in the brain systems that respond to these triggers [7]. Separate reviews of internet and gaming addiction, and of non-substance cue-reactivity, point in the same direction: the brain can learn to treat signals, images, and anticipation as part of the reward [8], [9].
This second figure shows the same prediction-error idea in table and graph form. It walks through baseline, cue, reward, and reward omission, and shows how dopamine activity shifts from the reward itself to the cue after learning.
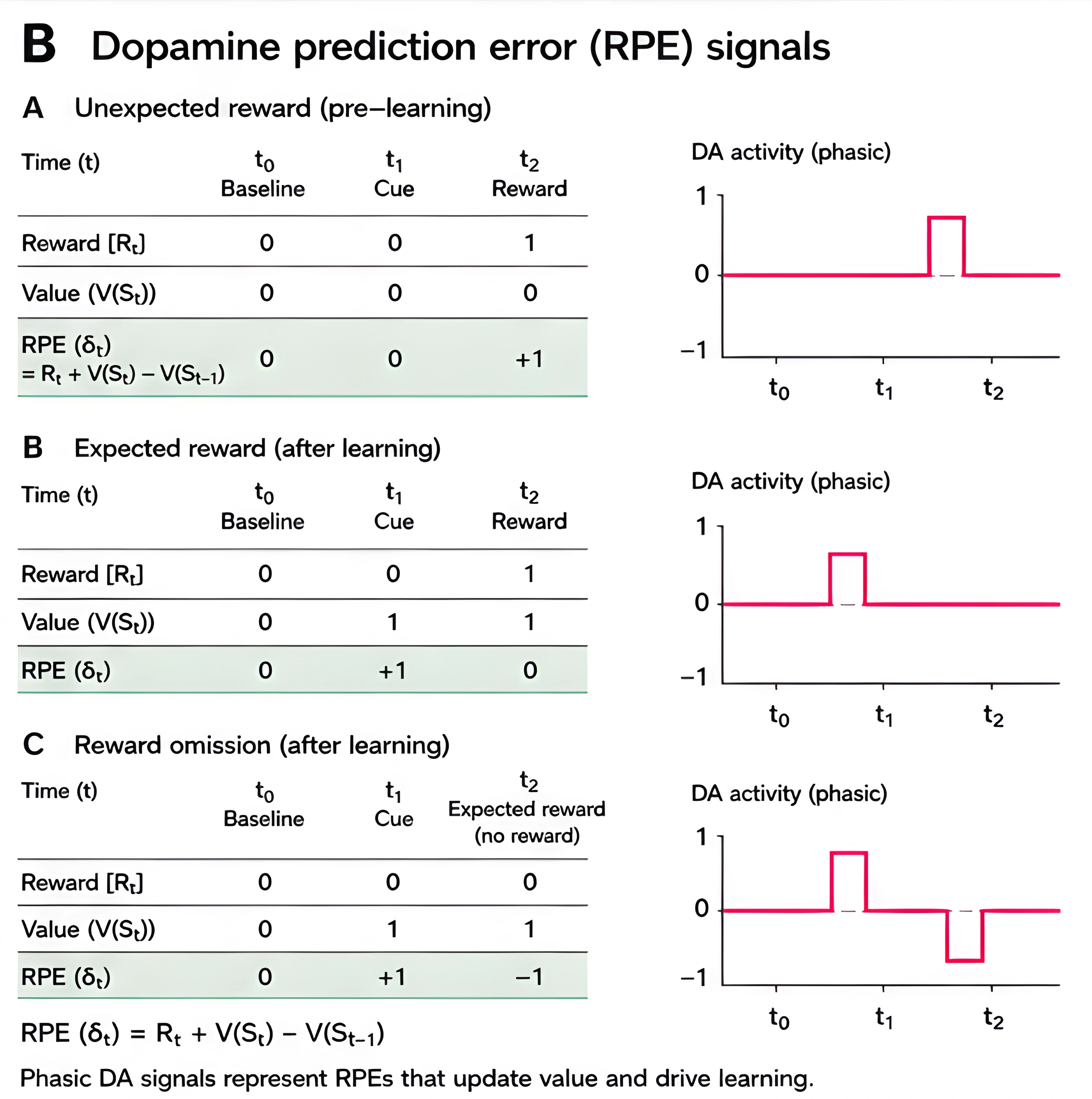
That matters for compulsive porn use, scrolling, gaming, and gambling because the cue is often what starts the loop. The person does not begin with pleasure. They begin with a trigger, a prediction, or a feeling state that the brain has learned to solve with the same behavior.
If "dopamine addiction" is not precise, what is the better model?
The better model is an overtrained reward loop.
Healthy rewards can activate the reward circuit too. Eating, socializing, sex, novelty, achievement, and music all do that. But addictive substances and highly stimulating behaviors can hit that system much harder. NIDA describes drug rewards as much larger than the smaller bursts linked to natural rewards [1], [2], [3]. Repeated exposure can then teach the brain to prioritize the addictive behavior over healthier goals [10]-[15], [21], [23].
That loop usually looks something like this:
With repetition, the loop gets easier to trigger and harder to interrupt.
This also helps explain something that confuses people in recovery: the reward often gets worse while the compulsion stays strong. A person may not even enjoy the behavior the way they once did, but the brain still reacts to the cues, the ritual, and the expectation. The pattern survives even after the original excitement has weakened [10], [11], [13], [21], [22].
That pattern is consistent with incentive-sensitization theory, where wanting can become stronger even when liking is weak or flat [10], [11], [13], [14]. It is also consistent with reward-learning models of addiction, where a high-value cue can keep driving behavior after the outcome stops delivering the same payoff [5], [11], [24].
The internet often responds to this with phrases like "dopamine detox," "dopamine reset," or "dopamine fasting." Those terms are catchy, but they can mislead people about what change actually involves.
The Harvard Health review makes the point clearly: a so-called dopamine fast does not literally lower your dopamine levels. You are not detoxing a natural brain chemical out of your system. The more charitable version of the idea is that you are stepping away from overstimulating cues and compulsive routines so you can regain some control [31], [34].
That distinction matters.
If a person thinks the goal is to drain dopamine, they are aiming at the wrong target. The better goal is to weaken automatic cue-response loops and create room for healthier patterns.
That can include:
Those strategies may help. But they work because they change learning, attention, and behavior over time, not because they "flush out" dopamine.
Recent reviews of dopamine fasting make the same point: the phrase is usually a sloppy label for cue reduction, habit interruption, and stimulus control, not a literal biochemical detox [31], [34]. The better language is behavioral retraining, not dopamine cleansing.
Another reason people believe they have a dopamine problem is that life can feel flatter after repeated overstimulation.
NIDA explains that with repeated drug exposure, the reward circuit can adapt. Sensitivity drops. The person may feel less pleasure from ordinary activities and more urgency around the addictive behavior. That does not mean the brain is permanently broken, but it does mean the person's reward system has been trained in an unhealthy direction [1], [2], [23], [24].
That flattening shows up in the addiction literature as anhedonia, reward dysregulation, and allostatic shift [10], [14]-[17], [25]-[28]. Reviews on drug addiction, anhedonia, and post-withdrawal reward processing describe a common pattern: ordinary rewards stop landing the way they used to, while cues, stress, and craving remain strong. The brain has not lost dopamine. It has adapted to a different reward environment [14]-[17], [25]-[29].
Clinical reviews also connect anhedonia with comorbid mood disorder, which matters because the flatness people feel in recovery is often mixed with depression, stress, or emotional exhaustion rather than being a pure dopamine deficit [26]. Post-withdrawal reward-processing studies likewise show that the brain can remain biased toward unhealthy reward cues even when ordinary reward processing is blunted [27].
This is why recovery often feels disappointing at first. Reading, walking, conversation, work, and sleep do not create the same immediate impact as the old behavior. Early recovery can feel slow, dull, or emotionally muted compared with the intense pattern the brain had learned to expect.
That experience is one reason people relapse. It is not only that they "want the high." Often they want relief from flatness, stress, irritability, or emptiness.
The same reward-learning logic applies to compulsive screen-bound sexual behavior, gaming, and other highly repetitive digital behaviors. Reviews of internet and videogame addiction describe reward-circuit involvement, impaired inhibitory control, and ventral striatal changes [8], [9], [17]-[19], [30]. Reviews of behavioral addictions and cue-reactivity also show that sexual cues, gambling cues, and other non-substance rewards can recruit overlapping neural substrates [6]-[9], [28], [29]. That is why modern platforms can feel so sticky: they provide repeated cues, intermittent reinforcement, and immediate novelty [8], [9], [33].
The newer public-facing article we reviewed, Wired for Want, is useful here as a contemporary example of the same pattern [33]. It is not a primary neuroscience study, but it captures a real behavioral phenomenon: modern systems can be engineered to keep the reward loop hot.
The practical conclusion is simple. If someone is stuck in porn, scrolling, gaming, or shopping loops, the problem is not that dopamine itself is "bad." The problem is that the brain has learned a cue-driven routine that keeps reopening the same pathway.
This third figure is a schematic of the core brain regions in that loop. It shows the prefrontal cortex, striatum, pallidum, thalamus, and VTA/SNc, and it makes the point that dopamine input is stronger to the striatum than to cortex.
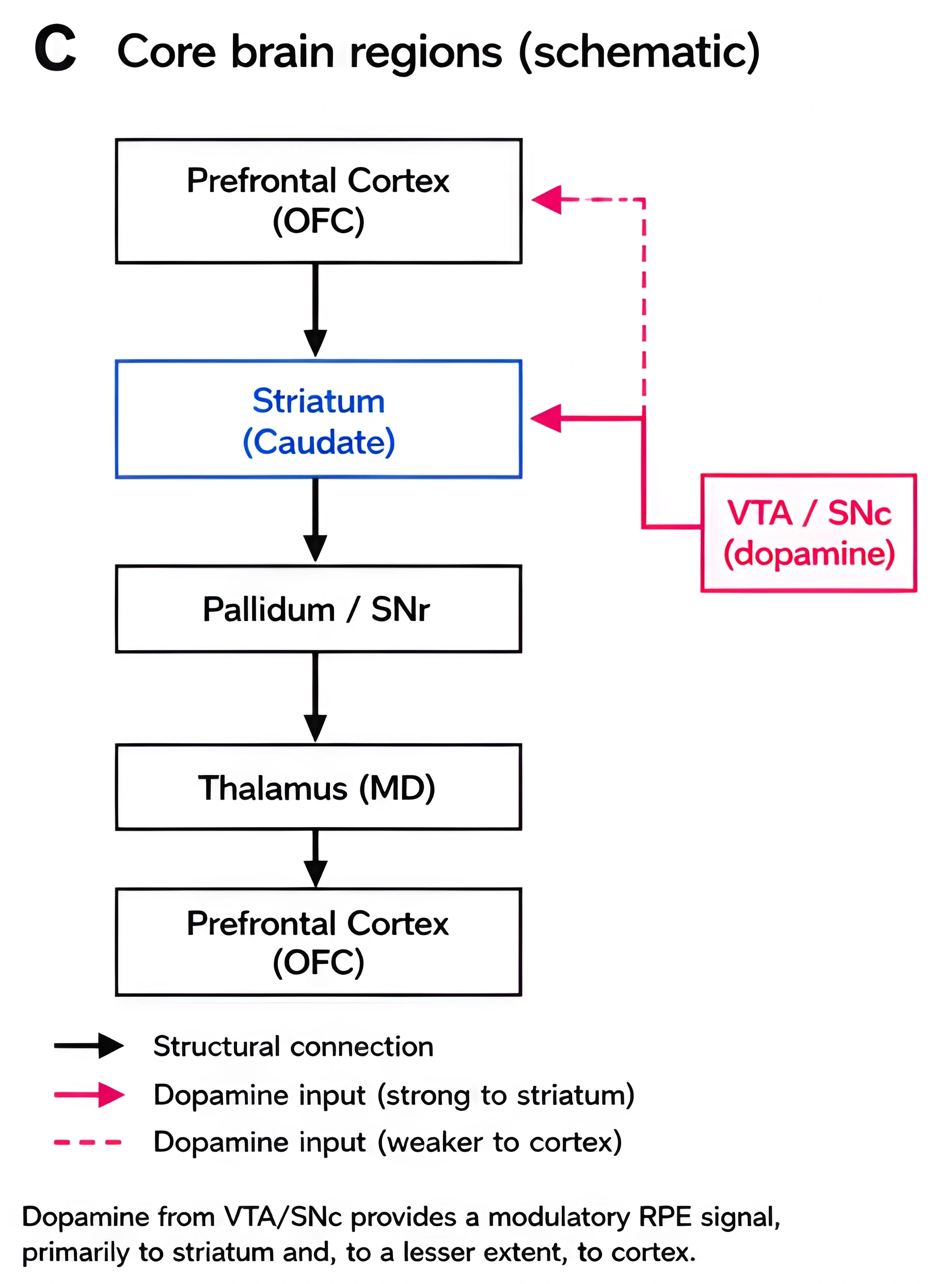
This fourth figure is an fMRI-style comparison from reversal learning. It shows lower activation in patients than controls across OFC, ventromedial striatum, caudate, and axial views, which matters because it suggests weaker flexibility in the circuit that should update behavior.
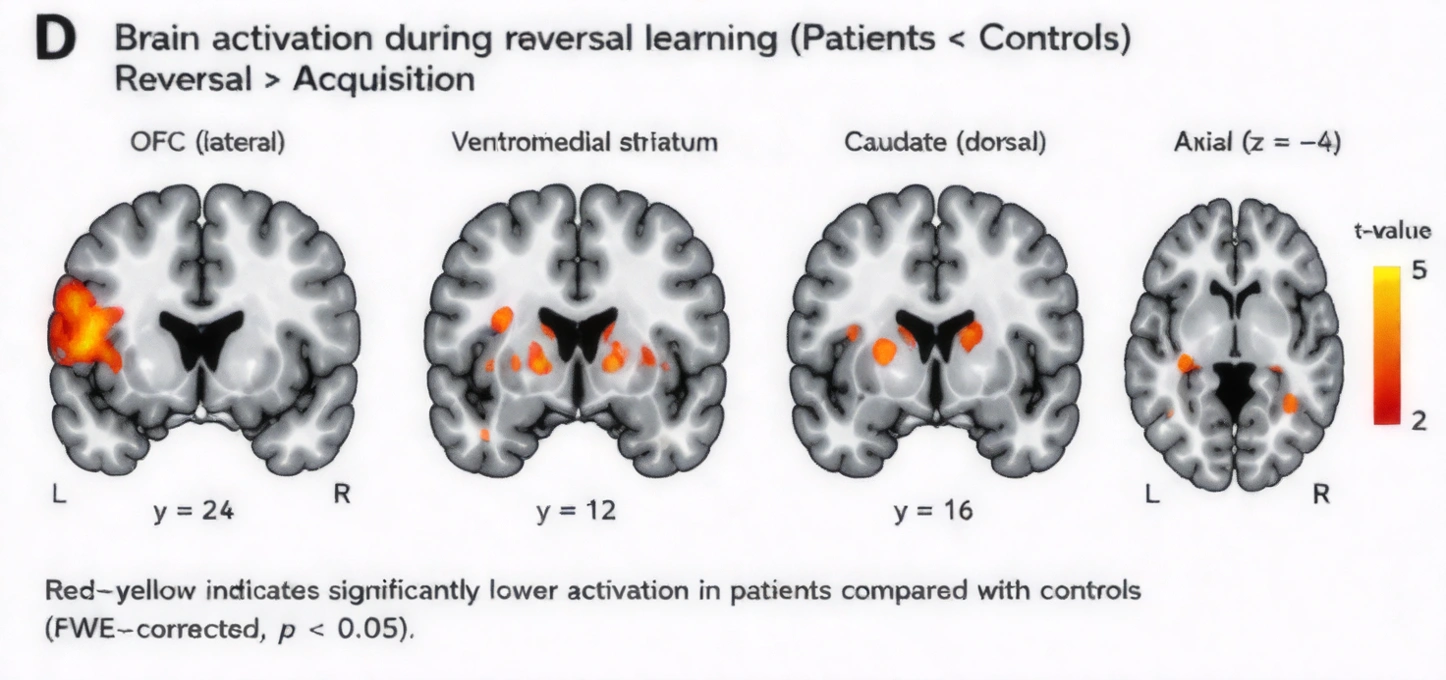
Assessment
This is the first question in our assessment. It is for people trying to leave pornography and related screen-based sexual behavior, and it helps identify what is still holding the behavior in place.
What does this behavior still give you that you don't want to lose?
If the real issue is not dopamine itself, then what should recovery focus on?
Recovery should focus on retraining the reward system.
That means targeting the habit machinery, not just trying to endure cravings. Reversal-learning research shows that flexibility depends heavily on frontocorticostriatal circuitry, while habit-formation reviews show that repeated reward can shift control toward automatic responding [18]-[21]. A 2015 circuit review and a 2021 mechanism paper make the same point from different angles: compulsivity is not just excess desire, but a control problem rooted in corticostriatal organization [17]-[19], [22]. Another paper in this source set shows that addiction-induced plasticity changes the underlying reward circuits in durable ways [23], [24].
Izquierdo and Jentsch frame reversal learning as a concrete way to measure impulsive and compulsive behavior in addiction, which is useful because recovery often fails when a person cannot flexibly switch away from the learned reward pattern [20].
That usually means working on several layers at once:
Notice what reliably starts the loop. Time, place, loneliness, conflict, devices, substances, fatigue, and secrecy all matter.
Change the sequence, not just the intention. If the same cue leads to the same ritual, insight alone usually will not be enough.
Rebuild healthier rewards that are slower but more stable: movement, sleep, social contact, meaningful work, sunlight, novelty, skill-building, and recovery structure.
Make the unwanted behavior harder to start. Remove saved payment methods, block sites, change routes, avoid using alone, or reduce access to high-risk environments.
If the pattern is severe, outside help matters. Treatment, therapy, structured recovery support, and accountability can do more than willpower alone. The treatment and implementation reviews in this source set point in the same direction: evidence-based care works best when it is structured, repeated, and supported by systems that make the healthy choice easier to sustain [32].
The phrase "dopamine addiction" is imprecise, but the behavior behind it can still be serious.
Take it seriously if:
That last point matters. A person may talk casually about dopamine, but some substance-related problems are not casual.
If you want a more accurate sentence, try this:
"My reward system has learned a pattern that keeps pulling me back, and I need to retrain that pattern."
That framing is less catchy than "dopamine addiction," but it is far more useful. It explains why cues matter, why cravings can outlast pleasure, why a dopamine detox is the wrong idea, and why recovery has to be practical, repetitive, and structured.
The goal is not to get rid of dopamine. The goal is to stop letting unhealthy loops dominate what your brain has learned to chase.
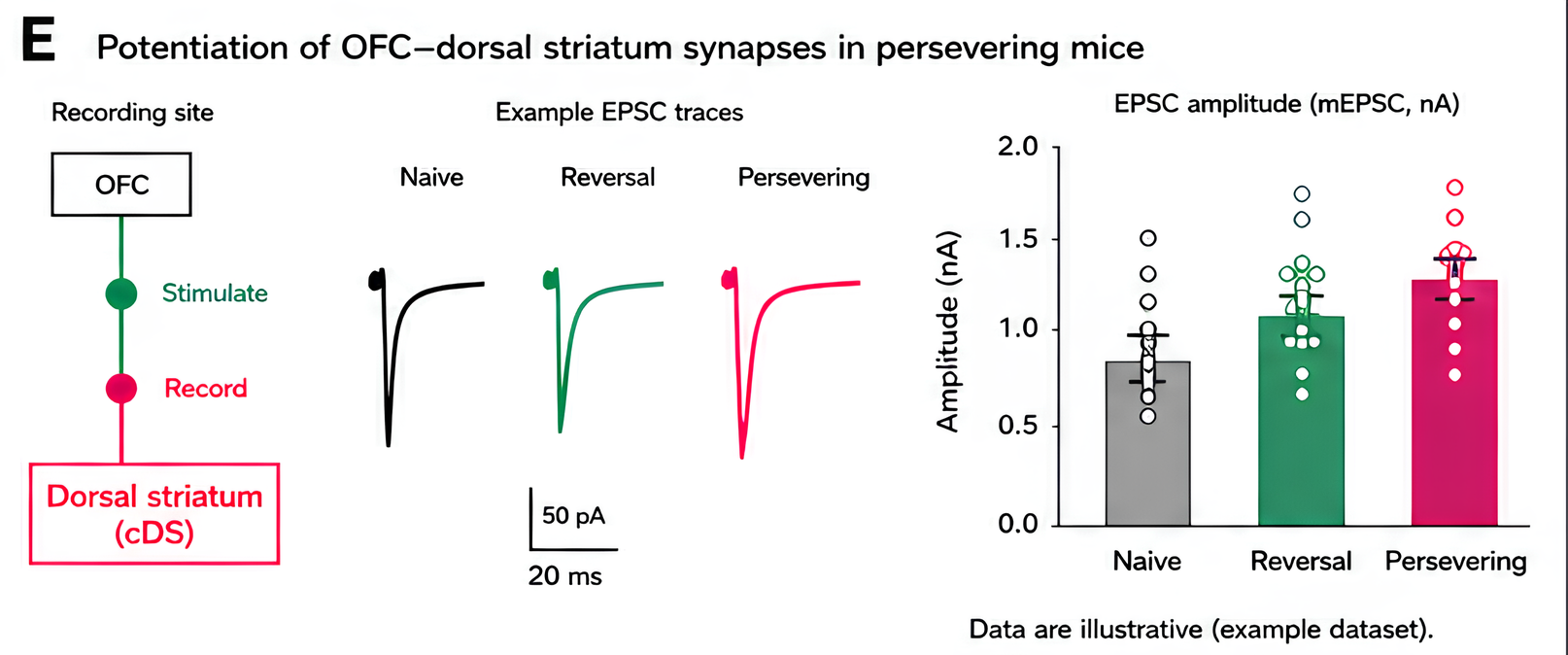
This fifth figure shows a mouse study of OFC-dorsal striatum synapses. It compares naive, reversal, and persevering animals and shows stronger synaptic responses in the persevering group, which supports the idea that repeated pattern-following can strengthen the old route.
This final composite combines the five source figures into one visual summary. It repeats the key circuit, prediction-error, and plasticity ideas in a single overview.
People ask "is dopamine addiction real" and "is dopamine addiction a real thing." The behavior is real, but the phrase is imprecise: what is happening is learned reward seeking, cue-reactivity, habit formation, and compulsivity [1], [10], [11].
Searches for "dopamine addiction symptoms" or "signs of dopamine addiction" usually point to the same pattern: compulsive checking, chasing stimulation, loss of control, craving when cues appear, and flattening of ordinary rewards [6], [10], [25].
If you are looking for "how to stop dopamine addiction," "how to overcome dopamine addiction," "how to beat dopamine addiction," "how to fix dopamine addiction," or "dopamine addiction treatment," the answer is not a magic reset. Help with dopamine addiction usually means cue control, habit interruption, therapy, and support [20], [32].
If you are searching for "how to do a dopamine detox," "how to do dopamine detox," or "dopamine detox plan," the better answer is that a dopamine reset is not a literal reset button. What people call "dopamine overload" is usually cue saturation, not too much dopamine that needs to be decreased or reduced globally. People sometimes ask to decrease dopamine or ask how to reduce dopamine, but that is the wrong target. A hard detox can also produce dopamine detox side effects such as irritability, rebound use, and all-or-nothing restriction [31], [34].
People search "can dopamine receptors heal," "can dopamine receptors be repaired," "how to heal dopamine receptors," "reset dopamine receptors," and "how to restore dopamine receptors." People also ask "can you fry your dopamine receptors." The better answer is that the brain is plastic: receptors and circuits can adapt and recover, but they are not literally fried hardware [23], [24].
There is no fixed timer for "how long for dopamine to reset," "how long until dopamine levels return to normal," "how to reset dopamine levels quickly," "how to reset dopamine levels," "how to reset dopamine baseline," "how to reset dopamine system," or "dopamine baseline recovery." Repeated overstimulation can also create a tolerance-like effect, which is why people search for dopamine tolerance. Recovery speed depends on the behavior, stress, sleep, and how much the loop is retrained [25]-[32].
Phrases like "dopamine obsession," "dopamine obsession in women," "chasing dopamine," "dopamine chase," "dopamine cycle," "dopamine dump," "dopamine junkie," "dopamine recovery," "how to recover from dopamine addiction," and even "addiction dopamine" are informal labels for the same cue-driven loop. They describe a learned pattern, not a formal diagnosis [10], [11], [13].
[1] National Institute on Drug Abuse, "Drugs, Brains, and Behavior: The Science of Addiction." Accessed: May 2, 2026. [Online]. Available: https://nida.nih.gov/publications/drugs-brains-behavior-science-addiction/drugs-brain
National Institute on Drug Abuse, "Drugs, Brains, and Behavior: The Science of Addiction." Baseline public explanation of addiction as a disorder of reward, stress, and self-control circuits; useful for the article's opening framing.
[2] National Institute on Drug Abuse, "Understanding Drug Use and Addiction DrugFacts." Accessed: May 2, 2026. [Online]. Available: https://nida.nih.gov/publications/drugfacts/understanding-drug-use-addiction
National Institute on Drug Abuse, "Understanding Drug Use and Addiction DrugFacts." Concise public overview that helps anchor the plain-language explanation of how repeated use changes reward learning and habit formation.
[3] G. Di Chiara and V. Bassareo, "Reward system and addiction: what dopamine does and does not do," Current Opinion in Pharmacology, vol. 7, pp. 69-76, 2007, doi: 10.1016/j.coph.2006.11.003.
G. Di Chiara and V. Bassareo, "Reward system and addiction: what dopamine does and does not do." Core review for distinguishing dopamine from simple pleasure and for explaining reinforcement rather than "dopamine as the addictive substance."
[4] J. H. Baik, "Dopamine signaling in reward-related behaviors," Frontiers in Neural Circuits, vol. 7, art. no. 152, 2013, doi: 10.3389/fncir.2013.00152.
J. H. Baik, "Dopamine signaling in reward-related behaviors." Useful receptor-level review for showing that dopamine signaling is not uniform and that D1-like and D2-like pathways play different roles in reward learning.
[5] R. Keiflin and P. H. Janak, "Dopamine prediction errors in reward learning and addiction: From theory to neural circuitry," Neuron, vol. 88, pp. 247-263, 2015, doi: 10.1016/j.neuron.2015.08.037.
R. Keiflin and P. H. Janak, "Dopamine prediction errors in reward learning and addiction: From theory to neural circuitry." Main source for the reward prediction error framework and the idea that dopamine functions as a teaching signal.
[6] K. Starcke, S. Antons, P. Trotzke, and M. Brand, "Cue-reactivity in behavioral addictions: A meta-analysis and methodological considerations," Journal of Behavioral Addictions, vol. 7, no. 2, pp. 227-238, 2018, doi: 10.1556/2006.7.2018.39.
K. Starcke, S. Antons, P. Trotzke, and M. Brand, "Cue-reactivity in behavioral addictions: A meta-analysis and methodological considerations." Best support for the claim that behavioral addictions show measurable cue-reactivity across subjective, physiological, and neural measures.
[7] H. R. Noori, A. C. Linan, and R. Spanagel, "Largely overlapping neuronal substrates of reactivity to drug, gambling, food and sexual cues: A comprehensive meta-analysis," European Neuropsychopharmacology, 2016, doi: 10.1016/j.euroneuro.2016.06.013.
H. R. Noori, A. C. Linan, and R. Spanagel, "Largely overlapping neuronal substrates of reactivity to drug, gambling, food and sexual cues: A comprehensive meta-analysis." Important for linking sexual cues, gambling cues, and drug cues to overlapping circuitry in the article's porn and screen-behavior section.
[8] A. Weinstein and M. Lejoyeux, "New developments on the neurobiological and pharmaco-genetic mechanisms underlying internet and videogame addiction," The American Journal on Addictions, vol. 24, pp. 117-125, 2015, doi: 10.1111/ajad.12110.
A. Weinstein and M. Lejoyeux, "New developments on the neurobiological and pharmaco-genetic mechanisms underlying internet and videogame addiction." Useful review for the screen-addiction section because it connects repetitive digital behavior to reward-circuit and inhibitory-control changes.
[9] R. F. Leeman and M. N. Potenza, "A targeted review of the neurobiology and genetics of behavioural addictions: An emerging area of research," Canadian Journal of Psychiatry, vol. 58, no. 5, pp. 260-273, 2013, doi: 10.1177/070674371305800503.
R. F. Leeman and M. N. Potenza, "A targeted review of the neurobiology and genetics of behavioural addictions: An emerging area of research." Broad behavioral-addiction review that helps justify treating gambling, gaming, buying, and similar compulsions as reward-system disorders.
[10] T. E. Robinson and K. C. Berridge, "The psychology and neurobiology of addiction: An incentive-sensitization view," Addiction, vol. 95, suppl. 2, pp. S91-S117, 2000, doi: 10.1046/j.1360-0443.95.8s2.19.x.
T. E. Robinson and K. C. Berridge, "The psychology and neurobiology of addiction: An incentive-sensitization view." Foundational theory source for craving, cue-triggered wanting, and the split between wanting and liking.
[11] B. J. Everitt and T. W. Robbins, "Neural systems of reinforcement for drug addiction: From actions to habits to compulsion," Nature Neuroscience, vol. 8, no. 11, pp. 1481-1489, 2005, doi: 10.1038/nn1579.
B. J. Everitt and T. W. Robbins, "Neural systems of reinforcement for drug addiction: From actions to habits to compulsion." Key review for the habit shift, showing how repeated reinforcement can move behavior from deliberate action toward compulsion.
[12] D. J. Nutt, A. Lingford-Hughes, D. Erritzoe, and P. R. A. Stokes, "The dopamine theory of addiction: 40 years of highs and lows," Nature Reviews Neuroscience, vol. 16, pp. 305-312, 2015, doi: 10.1038/nrn3939.
D. J. Nutt, A. Lingford-Hughes, D. Erritzoe, and P. R. A. Stokes, "The dopamine theory of addiction: 40 years of highs and lows." Good counterbalance source because it warns against a simplistic dopamine-only explanation.
[13] T. E. Robinson and K. C. Berridge, "Liking, wanting, and the incentive-sensitization theory of addiction," American Psychologist, vol. 71, no. 8, pp. 670-679, 2016, doi: 10.1037/amp0000059.
T. E. Robinson and K. C. Berridge, "Liking, wanting, and the incentive-sensitization theory of addiction." Best conceptual source for explaining why wanting can intensify even when liking is weak.
[14] T. E. Robinson and K. C. Berridge, "The incentive-sensitization theory of addiction 30 years on," Annual Review of Psychology, vol. 76, pp. 29-58, 2025, doi: 10.1146/annurev-psych-011624-024031.
T. E. Robinson and K. C. Berridge, "The incentive-sensitization theory of addiction 30 years on." Modern update that reinforces the long-term relevance of incentive-sensitization and its role in cue-driven addiction.
[15] M. Solinas, P. Belujon, P. O. Fernagut, M. Jaber, and N. Thiriet, "Dopamine and addiction: What have we learned from 40 years of research," Journal of Neural Transmission, vol. 126, pp. 481-516, 2019, doi: 10.1007/s00702-018-1957-2.
M. Solinas, P. Belujon, P. O. Fernagut, M. Jaber, and N. Thiriet, "Dopamine and addiction: What have we learned from 40 years of research." Broad synthesis that helps connect older dopamine findings with the current reward-learning model.
[16] R. A. Wise and C. J. Jordan, "Dopamine, behavior, and addiction," Journal of Biomedical Science, vol. 28, art. no. 83, 2021, doi: 10.1186/s12929-021-00779-7.
R. A. Wise and C. J. Jordan, "Dopamine, behavior, and addiction." Accessible review for linking dopamine to motivation, behavior, and addiction without reducing it to a pleasure chemical.
[17] C. L. Poisson, L. Engel, and B. T. Saunders, "Dopamine circuit mechanisms of addiction-like behaviors," Frontiers in Neural Circuits, vol. 15, art. no. 752420, 2021, doi: 10.3389/fncir.2021.752420.
C. L. Poisson, L. Engel, and B. T. Saunders, "Dopamine circuit mechanisms of addiction-like behaviors." Useful for the article's circuit-level explanation of addiction-like behavior in both substance and non-substance contexts.
[18] O. A. van den Heuvel et al., "Brain circuitry of compulsivity," European Neuropsychopharmacology, 2015, doi: 10.1016/j.euroneuro.2015.12.005.
O. A. van den Heuvel et al., "Brain circuitry of compulsivity." Important source for frontocorticostriatal compulsivity and impaired flexibility, which supports the recovery section.
[19] M. Figee et al., "Compulsivity in obsessive-compulsive disorder and addictions," European Neuropsychopharmacology, 2015, doi: 10.1016/j.euroneuro.2015.12.003.
M. Figee et al., "Compulsivity in obsessive-compulsive disorder and addictions." Helps support the claim that compulsivity overlaps across OCD and addiction and is not just a craving problem.
[20] A. Izquierdo and J. D. Jentsch, "Reversal learning as a measure of impulsive and compulsive behavior in addictions," Psychopharmacology, vol. 219, pp. 607-620, 2012, doi: 10.1007/s00213-011-2579-7.
A. Izquierdo and J. D. Jentsch, "Reversal learning as a measure of impulsive and compulsive behavior in addictions." Useful for explaining behavioral flexibility and why recovery often requires retraining switching and control.
[21] D. O'Tousa and N. Grahame, "Habit formation: Implications for alcoholism research," Alcohol, vol. 48, no. 4, pp. 327-335, 2014, doi: 10.1016/j.alcohol.2014.02.004.
D. O'Tousa and N. Grahame, "Habit formation: Implications for alcoholism research." Direct support for the habit-formation section and the claim that repeated reward can become automatic responding.
[22] M. Harada, V. Pascoli, A. Hiver, J. Flakowski, and C. Lüscher, "Corticostriatal activity driving compulsive reward seeking," Biological Psychiatry, vol. 90, pp. 808-818, 2021, doi: 10.1016/j.biopsych.2021.08.018.
M. Harada, V. Pascoli, A. Hiver, J. Flakowski, and C. Lüscher, "Corticostriatal activity driving compulsive reward seeking." Strong mechanistic paper for the role of corticostriatal plasticity in compulsive reward seeking.
[23] M. Kourosh-Arami, A. Komaki, and M. Gholami, "Addiction-induced plasticity in underlying neural circuits," Neurological Sciences, vol. 43, pp. 1605-1615, 2022, doi: 10.1007/s10072-021-05778-y.
M. Kourosh-Arami, A. Komaki, and M. Gholami, "Addiction-induced plasticity in underlying neural circuits." Useful for showing that repeated addiction can change underlying circuits in durable ways.
[24] C. Lüscher and P. H. Janak, "Consolidating the circuit model for addiction," Annual Review of Neuroscience, vol. 44, pp. 173-195, 2021, doi: 10.1146/annurev-neuro-092920-123905.
C. Lüscher and P. H. Janak, "Consolidating the circuit model for addiction." High-value synthesis for tying dopamine, reward circuitry, and addiction into one circuit model.
[25] D. S. Hatzigiakoumis, G. Martinotti, M. Di Giannantonio, and L. Janiri, "Anhedonia and substance dependence: Clinical correlates and treatment options," Frontiers in Psychiatry, vol. 2, art. no. 10, 2011, doi: 10.3389/fpsyt.2011.00010.
D. S. Hatzigiakoumis, G. Martinotti, M. Di Giannantonio, and L. Janiri, "Anhedonia and substance dependence: Clinical correlates and treatment options." Strong source for the article's discussion of flattened reward, anhedonia, and clinical recovery experience.
[26] M. Destoop, M. Morrens, V. Coppens, and G. Dom, "Addiction, anhedonia, and comorbid mood disorder: A narrative review," Frontiers in Psychiatry, vol. 10, art. no. 311, 2019, doi: 10.3389/fpsyt.2019.00311.
M. Destoop, M. Morrens, V. Coppens, and G. Dom, "Addiction, anhedonia, and comorbid mood disorder: A narrative review." Helpful for explaining that recovery flatness is often mixed with depression, stress, or mood symptoms.
[27] A. S. Huhn et al., "Evidence of anhedonia and differential reward processing in prefrontal cortex among post-withdrawal patients with prescription opiate dependence," Brain Research Bulletin, 2016, doi: 10.1016/j.brainresbull.2015.12.004.
A. S. Huhn et al., "Evidence of anhedonia and differential reward processing in prefrontal cortex among post-withdrawal patients with prescription opiate dependence." Supports the claim that ordinary reward processing can remain blunted after withdrawal.
[28] S. Liu et al., "Brain responses to drug cues predict craving changes in abstinent heroin users: A preliminary study," NeuroImage, 2021, doi: 10.1016/j.neuroimage.2021.118169.
S. Liu et al., "Brain responses to drug cues predict craving changes in abstinent heroin users: A preliminary study." Useful cue-reactivity source showing that cue brain responses can track craving change during abstinence.
[29] C. M. Olsen, "Natural rewards, neuroplasticity, and non-drug addictions," Neuropharmacology, vol. 61, no. 6, pp. 1109-1122, 2011, doi: 10.1016/j.neuropharm.2011.03.010.
Useful for showing that natural rewards also drive neuroplastic change, which helps place non-drug addictions inside the same broad learning framework.
[30] C. Chiamulera and R. J. West, "What role does dopamine really play in tobacco addiction?" Addiction, 2018, doi: 10.1111/add.14235.
C. Chiamulera and R. J. West, "What role does dopamine really play in tobacco addiction?" Useful cautionary review for keeping the dopamine story grounded in real addiction mechanisms rather than slogans.
[31] Y. Y. Fei, P. A. Johnson, N. A. L. Omran, A. Mardon, and J. C. Johnson, "Maladaptive or misunderstood? Dopamine fasting as a potential intervention for behavioral addiction," Lifestyle Medicine, vol. 3, art. no. e54, 2022, doi: 10.1002/lim2.54.
Y. Y. Fei, P. A. Johnson, N. A. L. Omran, A. Mardon, and J. C. Johnson, "Maladaptive or misunderstood? Dopamine fasting as a potential intervention for behavioral addiction." Best source for the article's critique of dopamine fasting as a literal biochemical detox.
[32] D. L. Sinclair et al., "Recovery-supportive interventions for people with substance use disorders: A scoping review," Frontiers in Psychiatry, vol. 15, art. no. 1352818, 2024, doi: 10.3389/fpsyt.2024.1352818.
D. L. Sinclair et al., "Recovery-supportive interventions for people with substance use disorders: A scoping review." Useful for the practical recovery section because it supports structured, supportive interventions over willpower-only advice.
[33] Z. Wyatt, "Wired for Want: How Dopamine Drives the New Epidemic of Everyday Addictions," Psychiatry and Behavioral Health, vol. 4, no. 1, pp. 1-6, 2025, doi: 10.33425/2833-5449.0018. Available: https://www.sciencexcel.com/article/wired-for-want-how-dopamine-drives-the-new-epidemic-of-everyday-addictions
Z. Wyatt, "Wired for Want: How Dopamine Drives the New Epidemic of Everyday Addictions." Contemporary public-facing piece that helps frame the article in modern attention and behavior patterns, but it is not primary evidence.
[34] Harvard Health, "Dopamine fasting: misunderstanding science spawns a maladaptive fad." Accessed: May 2, 2026. [Online]. Available: https://www.health.harvard.edu/blog/dopamine-fasting-misunderstanding-science-spawns-a-maladaptive-fad-2020022618917
Harvard Health, "Dopamine fasting: misunderstanding science spawns a maladaptive fad." Accessible public explainer that reinforces the article's point that dopamine fasting is usually a misunderstanding of brain science.
Choose the answer that fits best to see what is still holding the behavior in place.
Start assessmentNo account needed to start. Your answers are anonymous.